Ethanol Emulsion Test for Fats and Oils
The Ethanol Emulsion Test is a food test which determines the presence of a broad group of naturally occurring compounds known as lipids. Lipids consist of fats and oils. Other lipid tests include the Grease Spot Test and the Sudan Stain Test. The Grease spot test is performed on fats - lipids which are solid at room temperature. Sudan stain colours lipids red, but is a less common bench reagent than ethanol. The Ethanol Emulsion Test is the most common test amongst the three.
Procedure

Solid sample
Liquid sample
|
Observation
Solution remains colourless. No emulsion is formed.
A layer of cloudy white suspension forms at the top of the solution. (Upon close inspection you can see the tiny globules of fat suspended in the solution. This an emulsion. Foods with high lipid content have a ‘higher’ layer than foods with less). |
Interpretation
Lipids are not present
Lipids are present |
|
|
Principle of the Ethanol Emulsion Test
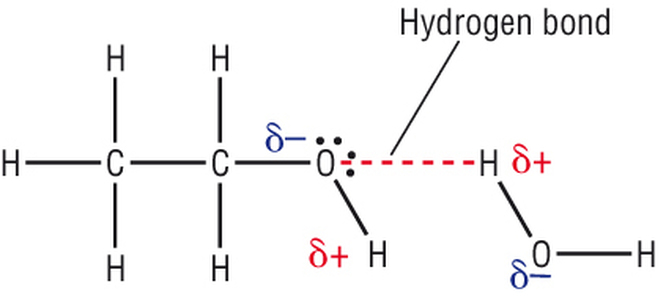
The solubilities of lipids and ethanol are exploited in this test.
Lipids are non-polar organic compounds. Hence they are soluble in organic solvents such as ethanol (alcohol), but insoluble in water.
Ethanol is an organic substance and so dissolves other organic substances; it is frequently used as an organic solvent.
However ethanol is also miscible in water due to the presence of the hydroxyl (-OH) functional groups and the shortness of its chain (2C). The hydroxyl group participates in hydrogen bonding with water.
The hydrophobic interaction of the carbon in the short chain with water is not great and is overcome by the hydrogen bonding.
Ethanol extracts the lipid from the crushed solid sample. As ethanol is miscible with lipids no change is seen upon its addition to the solid and liquid samples.
The lipid spontaneously comes out of solution when water is added and is dispersed as micelles (small droplets) throughout the solution of ethanol and water.( This happens as hydrophobic portion of the lipid molecules project inwards and excludes the aqueous environment; the hydrophilic portion (-COOH) group faces the aqueous environement.)
A layer is formed at the top as lipids are less dense than water. The droplets diffract light, appearing cloudy white.
CONCLUSIVE TEST
A positive test shows conclusively that lipids are present - and not the other major biological molecules.
1. Carbohydrates
3. Nucleic Acids – are insoluble in ethanol and soluble in water.
No change is seen in a negative test as there are no lipids to come out of solution.
Lipids are non-polar organic compounds. Hence they are soluble in organic solvents such as ethanol (alcohol), but insoluble in water.
Ethanol is an organic substance and so dissolves other organic substances; it is frequently used as an organic solvent.
However ethanol is also miscible in water due to the presence of the hydroxyl (-OH) functional groups and the shortness of its chain (2C). The hydroxyl group participates in hydrogen bonding with water.
The hydrophobic interaction of the carbon in the short chain with water is not great and is overcome by the hydrogen bonding.
Ethanol extracts the lipid from the crushed solid sample. As ethanol is miscible with lipids no change is seen upon its addition to the solid and liquid samples.
The lipid spontaneously comes out of solution when water is added and is dispersed as micelles (small droplets) throughout the solution of ethanol and water.( This happens as hydrophobic portion of the lipid molecules project inwards and excludes the aqueous environment; the hydrophilic portion (-COOH) group faces the aqueous environement.)
A layer is formed at the top as lipids are less dense than water. The droplets diffract light, appearing cloudy white.
CONCLUSIVE TEST
A positive test shows conclusively that lipids are present - and not the other major biological molecules.
1. Carbohydrates
- Reducing sugars and non- reducing sugars - slightly soluble in ethanol and soluble in water
- Starch – insoluble in both ethanol and water
3. Nucleic Acids – are insoluble in ethanol and soluble in water.
No change is seen in a negative test as there are no lipids to come out of solution.
Experimental Skills
The Ethanol Emulsion Test procedure is suitable for assessment of the Measurement and Manipulation Experimental skill.
Below are some criteria your teacher may use to assess you. He will be scrutinizing the steps you take to prevent contamination , including cross contamination from other food samples.
CRITERIA
Preparation of a Solid Sample:
Below are some criteria your teacher may use to assess you. He will be scrutinizing the steps you take to prevent contamination , including cross contamination from other food samples.
CRITERIA
Preparation of a Solid Sample:
- Completely crushes adequate amount of food with a clean and dry mortar and pestle.
- Transfers an adequate amount of crushed food sample to a clean test tube.
- Adds 2cm3 ethanol above the food sample.
- Shakes the test tube thoroughly and safely (using a vortex mixer if available).
- Allows the solid to settle for about 3 min.
- Decants the ethanol carefully, into a clean, dry test tube without transference of food particles.
- Adds 2cm3 of de-ionized water to the test tube.
- Compares each result with the negative control.
- Compares the heights of the emulsion layer of all the samples.
Discussion Questions
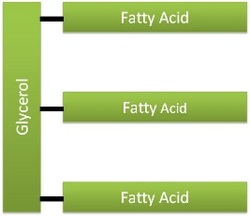
Triglyceride Structure
What are Lipids?
Lipids are a class of organic compounds consisting of Carbon, Hydrogen and Oxygen; their Hydrogen: Oxygen ratio is greater than 2:1. Lipids are non - polar compounds, meaning they are soluble in organic solvents (e.g. ethanol) and insoluble in aqueous solvents (e.g. water).
Most Dietary Lipids are triglycerides. Describe the structure of the triglycerides.
Triglycerides consist of a glycerol backbone bonded to 3 fatty acid chains of varying lengths.
Account for the physical states of Fats and Oils.
Fats are triglycerols with long chain fatty acids. Van der Waals interactions between the long chains give rise to high melting points. Hence, fats are solid at room temperature.Oils are trigycerols with shorter fatty acid chains. As the Van der Waals interactions are not great, oils have a lower melting point and are liquid at room temperature.
Outline the function of dietary lipids in the body. Name 3 sources of dietary lipids.
Lipds obtained from the diet function as energy storage molecules, structural components of the phospholipid bilayer of cell membranes and cell signaling molecules. Layers of fat beneath the skin serve as insulation and around organs as protection from mechanical injuries. Fat soluble Vitamins A, D,E and K are stored in adipose ( fatty) tissue. Oily secretions (sebum) protect the skin from drying out.Food sources of lipids include walnuts, wheat germ, coconut oil and palm kernel oil, cheese and butter.
Why is a fat-free diet unadvisable?
Persons who have a fat-deficient diet are also deficient in fat-soluble vitamins. ( A, D,E and K). Symptoms include blurred vision, weak bones – movement of calcium into soft tissues, digestive tract problems and numerous skin, hair and nail problems. A deficiency of Omega 3 essential fatty acids increases the risk for certain cancers – breast, prostate and colon, and also for diabetes.
Define obesity. Name three diseases associated with obesity.
Obesity is the excessive accumulation of body fat which poses a risk to health. Three diseases associated with obesity are Coronary Heart Disease, Type II Diabetes and certain types of cancers – endometrial, breast and colon.
What is the reason for crushing the soild food sample and leaving it to stand in ethanol for a few minutes?
Breaking the cells exposes the lipids stored inside so that they can be extracted by the ethanol solvent.
Lipids are a class of organic compounds consisting of Carbon, Hydrogen and Oxygen; their Hydrogen: Oxygen ratio is greater than 2:1. Lipids are non - polar compounds, meaning they are soluble in organic solvents (e.g. ethanol) and insoluble in aqueous solvents (e.g. water).
Most Dietary Lipids are triglycerides. Describe the structure of the triglycerides.
Triglycerides consist of a glycerol backbone bonded to 3 fatty acid chains of varying lengths.
Account for the physical states of Fats and Oils.
Fats are triglycerols with long chain fatty acids. Van der Waals interactions between the long chains give rise to high melting points. Hence, fats are solid at room temperature.Oils are trigycerols with shorter fatty acid chains. As the Van der Waals interactions are not great, oils have a lower melting point and are liquid at room temperature.
Outline the function of dietary lipids in the body. Name 3 sources of dietary lipids.
Lipds obtained from the diet function as energy storage molecules, structural components of the phospholipid bilayer of cell membranes and cell signaling molecules. Layers of fat beneath the skin serve as insulation and around organs as protection from mechanical injuries. Fat soluble Vitamins A, D,E and K are stored in adipose ( fatty) tissue. Oily secretions (sebum) protect the skin from drying out.Food sources of lipids include walnuts, wheat germ, coconut oil and palm kernel oil, cheese and butter.
Why is a fat-free diet unadvisable?
Persons who have a fat-deficient diet are also deficient in fat-soluble vitamins. ( A, D,E and K). Symptoms include blurred vision, weak bones – movement of calcium into soft tissues, digestive tract problems and numerous skin, hair and nail problems. A deficiency of Omega 3 essential fatty acids increases the risk for certain cancers – breast, prostate and colon, and also for diabetes.
Define obesity. Name three diseases associated with obesity.
Obesity is the excessive accumulation of body fat which poses a risk to health. Three diseases associated with obesity are Coronary Heart Disease, Type II Diabetes and certain types of cancers – endometrial, breast and colon.
What is the reason for crushing the soild food sample and leaving it to stand in ethanol for a few minutes?
Breaking the cells exposes the lipids stored inside so that they can be extracted by the ethanol solvent.