|
|
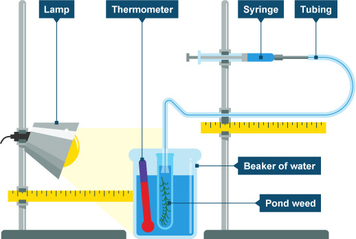
Effect of Light Intensity
|
|
|
3. Suggest how this procedure can be modified so that the distance between the lamp and the plant is not varied.
The power of the lamp may be varied e.g. use of 20W, 40W, 60W and 100W bulbs. 4. Why was the laboratory darkened? The laboratory was darkened to reduce light from other sources, to ensure that the light intensity was mainly due to the lamp. |
5. What are the main sources of inaccuracy in the experiment?
When counting bubbles, the bubbles may be of varied sizes, leading to inaccurate conclusions about the rate of oxygen production. If the rate of bubbling is very high, then there may be errors in counting. Another source of inaccuracy is the presence of other light sources, of which intensities may be varied by shifting shadows.
5. How can we prove that oxygen was the gas produced?
A glowing splint should be placed near the mouth of the test tube, where the gas was collected.If oxygen is present the splint is re-ignited into a flame.
6. If the gas which was produced was analyzed and found not to be pure oxygen, how would you account for this?
Water contains other dissolved gases which came out of solution and mixed with the oxygen. Carbon dioxide is expected in this mixture, particularly if sodium hydrogen carbonate was used.
7. What is the relationship between the light intensity and the rate of photosynthesis at low light intensities?
At low light intensities, the rate of photosynthesis increases with the intensity; the rate is directly proportional to intensity ( but inversely proportional to distance)
8. Why does the rate of photosynthesis not increase at very high light intensities?
At very high light intensities, the rate of photosynthesis levels off or remains constant. At this point, another factor - other than light - contributing to the rate of photosynthesis is limiting. The most likely reason for the leveling off of the reaction rate is the saturation of active sites in the enzymes catalyzing the reactions.
In the case of this experiment - the limiting factors are unlikely to be water or carbon dioxide. A limiting factor may be temperature - a slight increase in the ambient temperature may result an increase in the rate of reaction. However, a high temperatures the enzymes catalyzing the photosynthetic reactions would be denatured.
When counting bubbles, the bubbles may be of varied sizes, leading to inaccurate conclusions about the rate of oxygen production. If the rate of bubbling is very high, then there may be errors in counting. Another source of inaccuracy is the presence of other light sources, of which intensities may be varied by shifting shadows.
5. How can we prove that oxygen was the gas produced?
A glowing splint should be placed near the mouth of the test tube, where the gas was collected.If oxygen is present the splint is re-ignited into a flame.
6. If the gas which was produced was analyzed and found not to be pure oxygen, how would you account for this?
Water contains other dissolved gases which came out of solution and mixed with the oxygen. Carbon dioxide is expected in this mixture, particularly if sodium hydrogen carbonate was used.
7. What is the relationship between the light intensity and the rate of photosynthesis at low light intensities?
At low light intensities, the rate of photosynthesis increases with the intensity; the rate is directly proportional to intensity ( but inversely proportional to distance)
8. Why does the rate of photosynthesis not increase at very high light intensities?
At very high light intensities, the rate of photosynthesis levels off or remains constant. At this point, another factor - other than light - contributing to the rate of photosynthesis is limiting. The most likely reason for the leveling off of the reaction rate is the saturation of active sites in the enzymes catalyzing the reactions.
In the case of this experiment - the limiting factors are unlikely to be water or carbon dioxide. A limiting factor may be temperature - a slight increase in the ambient temperature may result an increase in the rate of reaction. However, a high temperatures the enzymes catalyzing the photosynthetic reactions would be denatured.