What is Benedict's Test for non-reducing sugars?
Benedict's Test for non-reducing Sugars is a test which determines the presence of non-reducing sugars in a test solution.
The principal reagent in Benedict's Test for Reducing Sugars is Benedict's Solution which contains
What are non-reducing sugars?
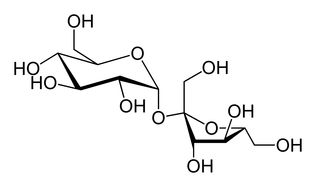
Non-reducing sugars are sugars which do not have an aldehyde functional group - the reducing species. As non-reducing sugars do not have the aldehyde group, they cannot reduce copper (I) (blue) to the copper(II) (red).
Sucrose is the most common disaccharide non-reducing sugar. Many students confuse the Benedict's Test for Non-Reducing Sugars with the Benedict's Test for Reducing Sugars, perhaps because some of the steps in the procedure are similar.
|
|
|
PROCEDURE
What is the procedure for the Benedict's Test for Reducing Sugars?
A liquid food sample does not need prior preparation except dilution if viscous or concentrated.
For a solid sample prepare a test solution by crushing the food and adding a moderate amount of distilled water. Decant the suspension to remove large particles. Use the decanted liquid as the test solution.
For a solid sample prepare a test solution by crushing the food and adding a moderate amount of distilled water. Decant the suspension to remove large particles. Use the decanted liquid as the test solution.
- Add 2 cm3 of the sample solution to a test tube.
- Add 1 cm3 of dilute hydrochloric acid and boil for one minute.
- Allow the tube to cool and then neutralize the acid with sodium hydrogen carbonate. Exercise caution due to 'fizzing' or effervescence.
Check with blue litmus paper or pH paper. Do not worry if the resulting solution is slightly alkaline; Benedict's solution is also alkaline since an alkaline medium in needed for oxidation of the copper (II) ions by the sugars
- Leave the test tube in a boiling water bath for about 5 minutes, or until the colour of the mixture does not change.
- Observe the colour changes during that time as well as the final colour.
- To prepare a control, repeat the steps using 2 cm3 of distilled water instead of sample solution.
OBSERVATIONS/RESULTS
What are the expected results of Benedict's Test for non-reducing sugars?
|
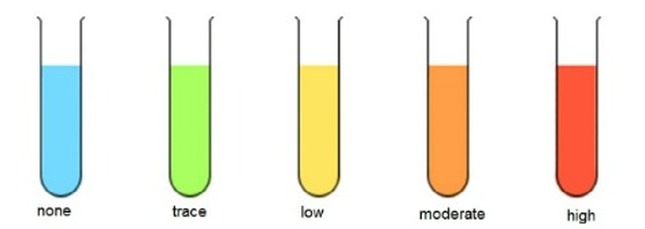
Observation
No Colour Change (Blue) Green Yellow Orange Brick-red |
Interpretation
No non-reducing sugars present Trace amounts of non-reducing sugars present Low amounts of reducing sugars present Moderate amounts of reducing sugars present Large amounts of non-reducing sugars present |
What results are expected if a food sample has already been tested for reducing sugars?
The test for non-reducing sugars is often conducted on a food sample which tested negative for reducing sugar.
If reducing sugars have been shown to be present, a heavier precipitate is often observed when the test for non-reducing sugar is conducted.
DISCUSSION
What is the principle of the Benedict's Test for non-reducing Sugars?
Disaccharides are hydrolyzed to their constituent monosaccharides when boiled in dilute hydrochloric acid.
The monosaccharide products of hydrolysis are reducing sugars i.e. have the aldehyde functional group and can reduce copper in the presence of alkali producing the colour changes.
The monosaccharide products of hydrolysis are reducing sugars i.e. have the aldehyde functional group and can reduce copper in the presence of alkali producing the colour changes.
Why was sodium hydrogen carbonate added to the mixture?
Neutralization. Sodium hydrogen carbonate is necessary for neutrallization as the reduction of the copper(II) ions will not take place in acidic conditions - of excess acid is present.
State the role of copper sulphate in Benedict's Solution.
Reduced Species. The blue copper(II) ions from copper(II) sulphate are reduced to red copper(I) ions by the aldehyde groups in the reducing sugars. This accounts for the colour changes observed.
The red copper(I) oxide formed is insoluble in water and is precipitated out of solution. This accounts for the precipitate formed.
As the concentration of reducing sugar increases, the nearer the final colour is to brick-red and the greater the precipitate formed.
The red copper(I) oxide formed is insoluble in water and is precipitated out of solution. This accounts for the precipitate formed.
As the concentration of reducing sugar increases, the nearer the final colour is to brick-red and the greater the precipitate formed.
State the role of sodium sodium carbonate in Benedict's Solution.
Alkalinization. Sodium carbonate provides the alkaline conditions which are required for the redox reaction above.
State the role of sodium citrate in Benedict's Solution.
Stabilization. Sodium citrate complexes with the copper (II) ions so that they do not deteriorate to copper(I) ions during storage.
Are there alternative tests to Benedict's Test for Reducing Sugars?
The Fehling's Test for non-reducing sugar is an alternative to the Benedict's Test. However it is less popular as it less sensitive and requires that the reagents - Fehling's solutions A and B - be kept separate until the experiment is carried out.
|
|